Featured Scientist

Huai-Jen Tsai
College of Science and Engineering
Huai-Jen Tsai
Department of Life Science,
Fu Jen Catholic University, New Taipei, Taiwan.
Article published in
"EMBO Journal" 2021 Jun 1;40(11):e104123
Poly(U)-specific endoribonuclease ENDOU promotes translation of human CHOP mRNA by releasing uORF element-mediated inhibition
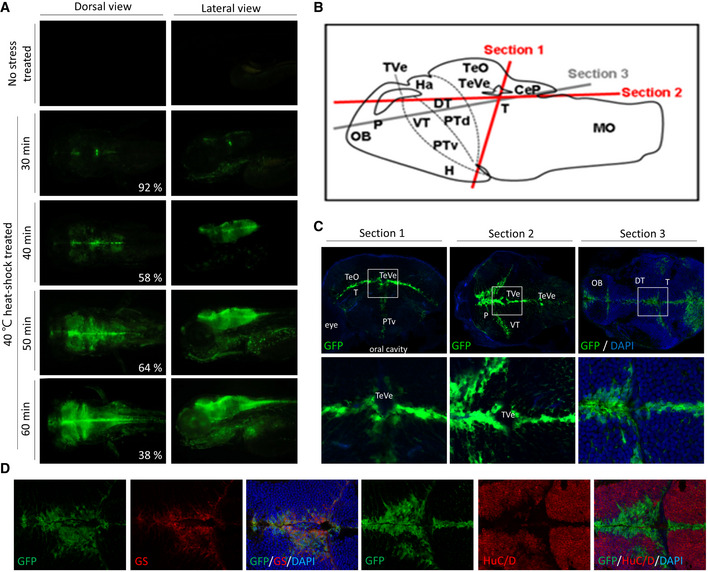
Upstream open reading frames (uORFs) are known to negatively affect translation of the downstream ORF. The regulatory proteins involved in relieving this inhibition are however poorly characterized. In response to cellular stress, eIF2α phosphorylation leads to an inhibition of global protein synthesis, while translation of specific factors such as CHOP is induced. We analyzed a 105-nt inhibitory uORF in the transcript of human CHOP (huORFchop) and found that overexpression of the zebrafish or human ENDOU poly(U)-endoribonuclease (Endouc or ENDOU-1, respectively) increases CHOP mRNA translation also in the absence of stress. We also found that Endouc/ENDOU-1 binds and cleaves the huORFchop transcript at position 80G-81U, which induces CHOP translation independently of phosphorylated eIF2α. However, both ENDOU and phospho-eIF2α are nonetheless required for maximal translation of CHOP mRNA. Increased levels of ENDOU shift a huORFchop reporter as well as endogenous CHOP transcripts from the monosome to polysome fraction, indicating an increase in translation. Furthermore, we found that the uncapped truncated huORFchop-69-105-nt transcript contains an internal ribosome entry site (IRES), facilitating translation of the cleaved transcript. Therefore, we propose a model where ENDOU-mediated transcript cleavage positively regulates CHOP translation resulting in increased CHOP protein levels upon stress. Specifically, CHOP transcript cleavage changes the configuration of huORFchop thereby releasing its inhibition and allowing the stalled ribosomes to resume translation of the downstream ORF.
Keywords: CHOP; Zebrafish Endouc; human ENDOU-1; translational control; uORF.
55 views