Featured Scientist

Ying-Hung Lin, Ph.D.
Associate Professor
Graduate Institute of Biomedical and Pharmaceutical Science
Education & Academic Qualifications
Ph.D., Graduate Institute of Basic Sciences, National Cheng Kung University, College of Medicine, Taiwan
Research Interests
Molecular and Cellular Biology, Andrology, Spermatogenesis
Genetics of Male Infertility
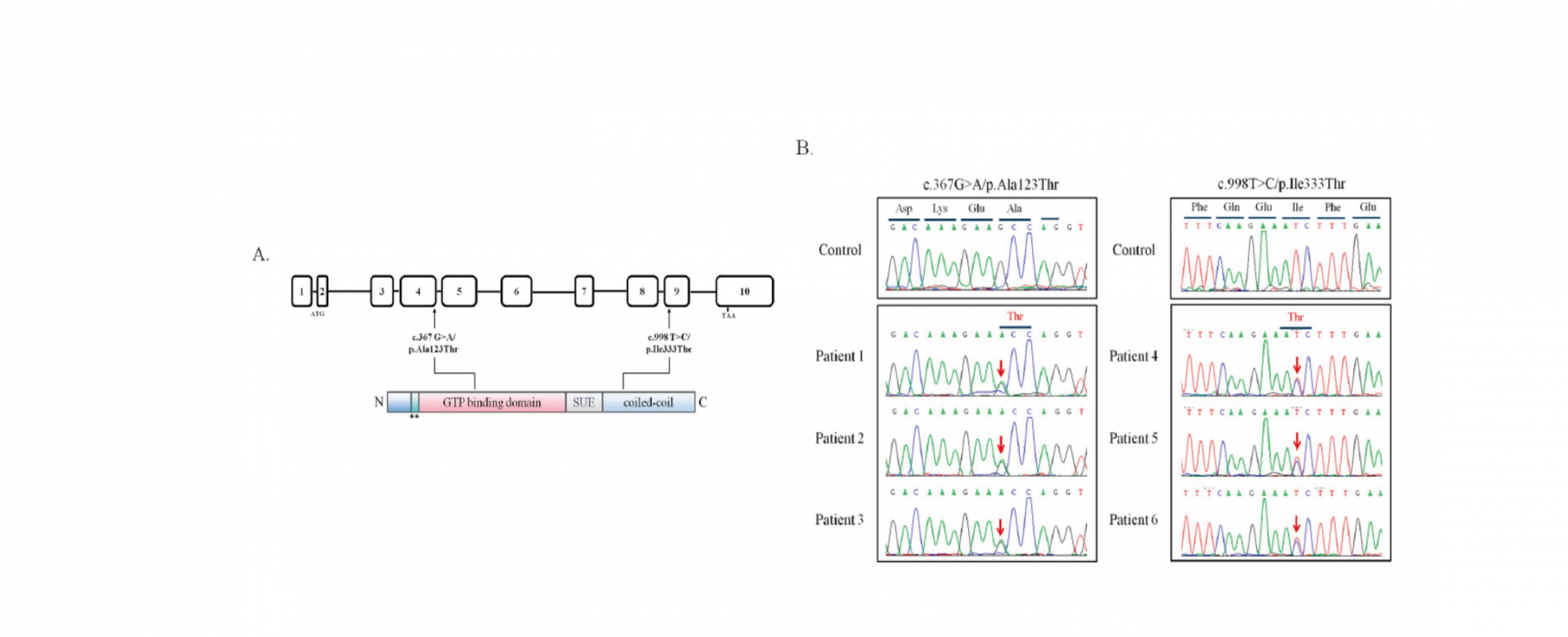
Infertility has been documented as a global public health concern by the World Health Organization (WHO), and approximately 9% of couples are affected by infertility worldwide. Male infertility accounts for 30% of couple infertility cases and affects approximately 7% of all men. One of the major causes of male infertility is teratozoospermia, which is defined as abnormal morphology in over 85% of sperm. It is frequently accompanied with sperm DNA defects. Moreover, sperm DNA damage limits the possibility of achieving successful pregnancy outcomes through assisted reproductive technology. In addition, sperm with high levels of DNA damage or abnormal DNA packaging has negative effects on embryo development and pregnancy outcomes, including pregnancy loss, recurrent spontaneous abortion, and lower live birth rates. To date, several mutations in certain genes have been linked to sperm DNA damage in a clinical aspect (i.e. PROTAMINE, AURKC, SPATA16, PICK1, DPY19L2, and SEPT12). SEPT14 was originally identified as a novel testis-expressed protein. In male reproduction, the expressional levels of SEPT14 transcripts are required for human spermatogenesis through evaluating the testicular tissues of men with normal and defective, including hypospermatogenesis, maturation arrest, and sertoli cell–only syndrome . Moreover, SEPT14 is mainly localized in the front of the acrosome, neck, and tail. The main objective of this study was to evaluate the potential genetic effects of SEPT14 on male infertility through sequencing the SEPT14 coding region.
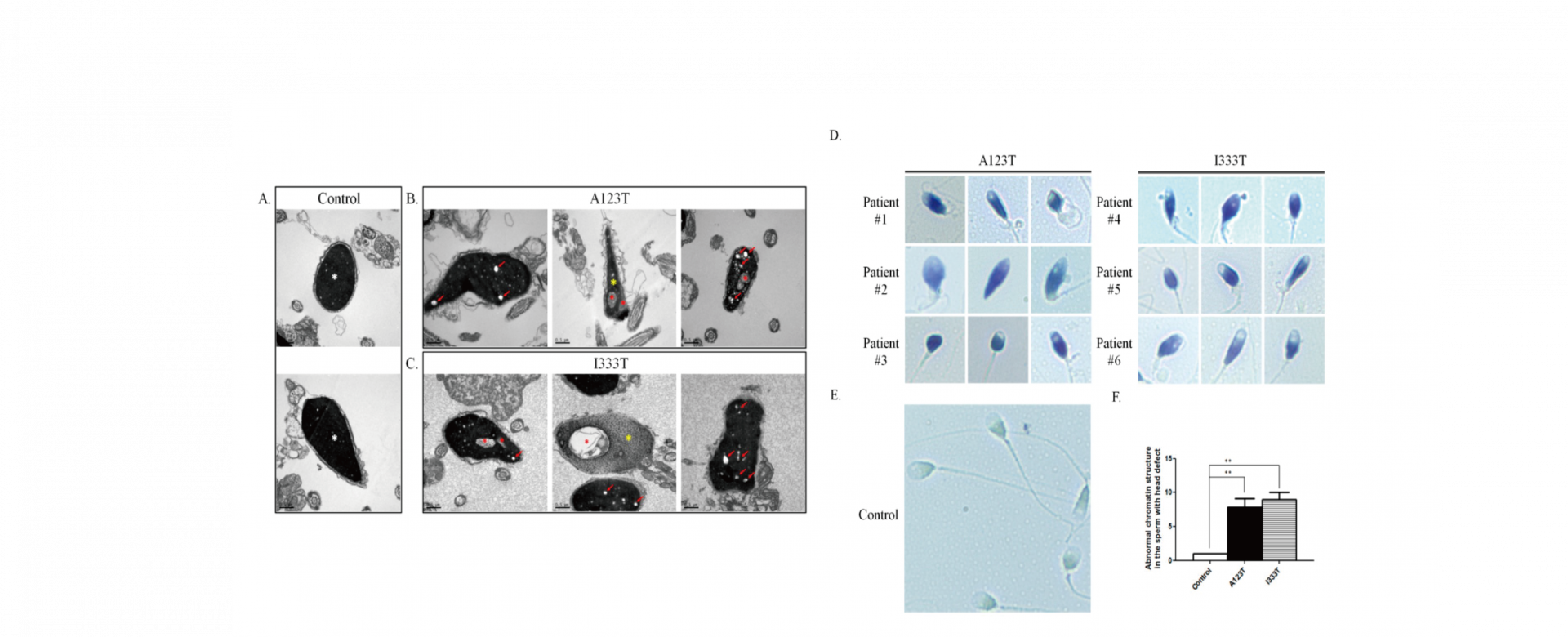
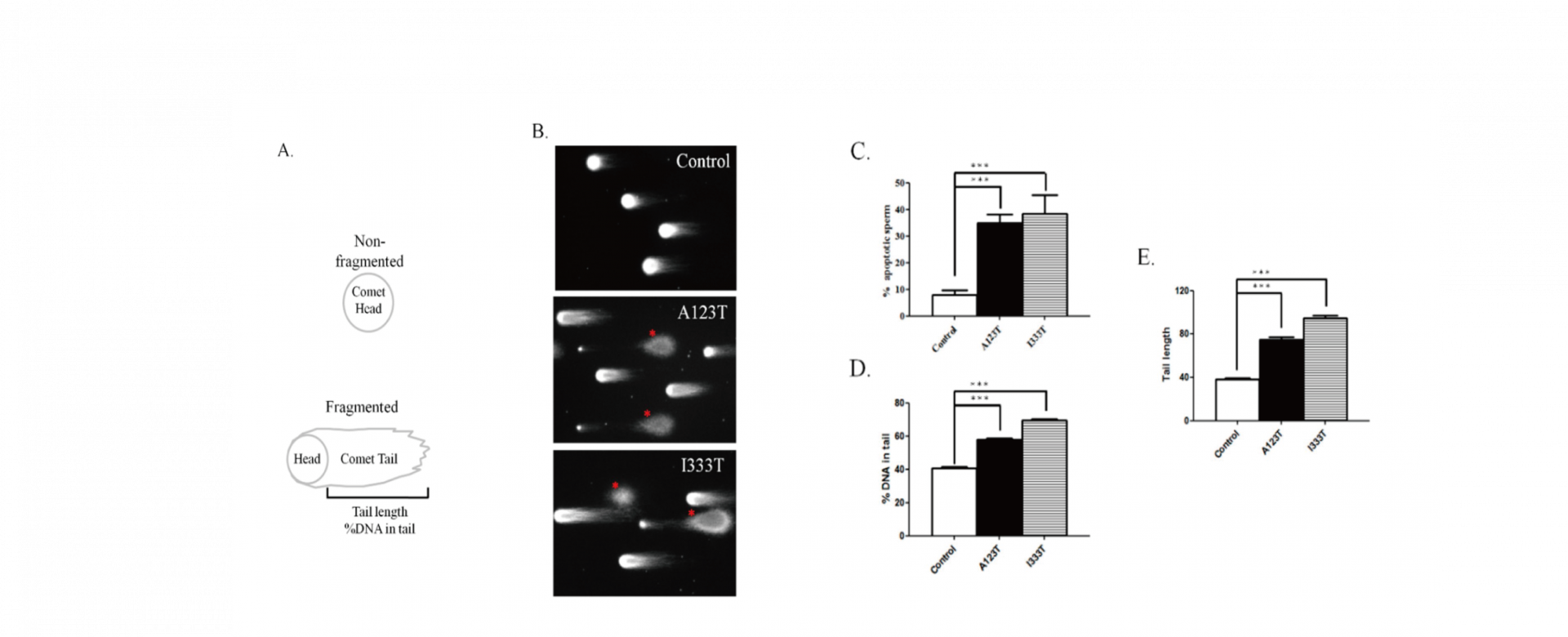
To address this research gap, 254 men with sperm abnormalities and 116 normozoospermic men were recruited, and the whole-coding regions of SEPT14 were sequenced. Two heterozygous mutations, p.Ala123Thr (3/254 vs. 0/116) and p.Ile333Thr (3/254 vs. 0/116), were identified in these cases. A high percentage of defective sperm heads was found in sperm with mutated SEPT14. Both mutations are highly evolutionarily conserved among vertebrates. The results of a fine morphological and chromatin structural analysis indicated severely malformed sperm heads with abnormal chromatin packaging through transmission electron microscopy and Toluidine blue staining. Compared with controls, high DNA fragmentation was demonstrated in sperm from cases carrying SEPT14 mutations using the comet assay. In addition, these two mutations in SEPT14 affected its polymerization ability in vitro. These data revels that the two SEPT14 missense mutations impaired sperm head morphology and induced DNA damage. Our study suggests that genetic variant of SEPT14 is one of the effects for human sperm formation and male fertility.
118 views