Featured Scientist

Jung-Feng Hsieh, Ph.D.
Professor
Food Proteomics
Proteomics is a powerful method that can simultaneously analyze several hundred proteins in complex mixtures through the use of high-resolution two-dimensional gel electrophoresis coupled with mass spectrometry.
Proteomics approaches have been used to investigate milk proteins.
Milk, which contains 3.3% protein, is a valuable nutrient source for humans.
Approximately 80% of milk protein is composed of caseins, and 20% is composed of whey proteins, including the well-known β-lactoglobulin, α-lactalbumin, and milk serum proteins.
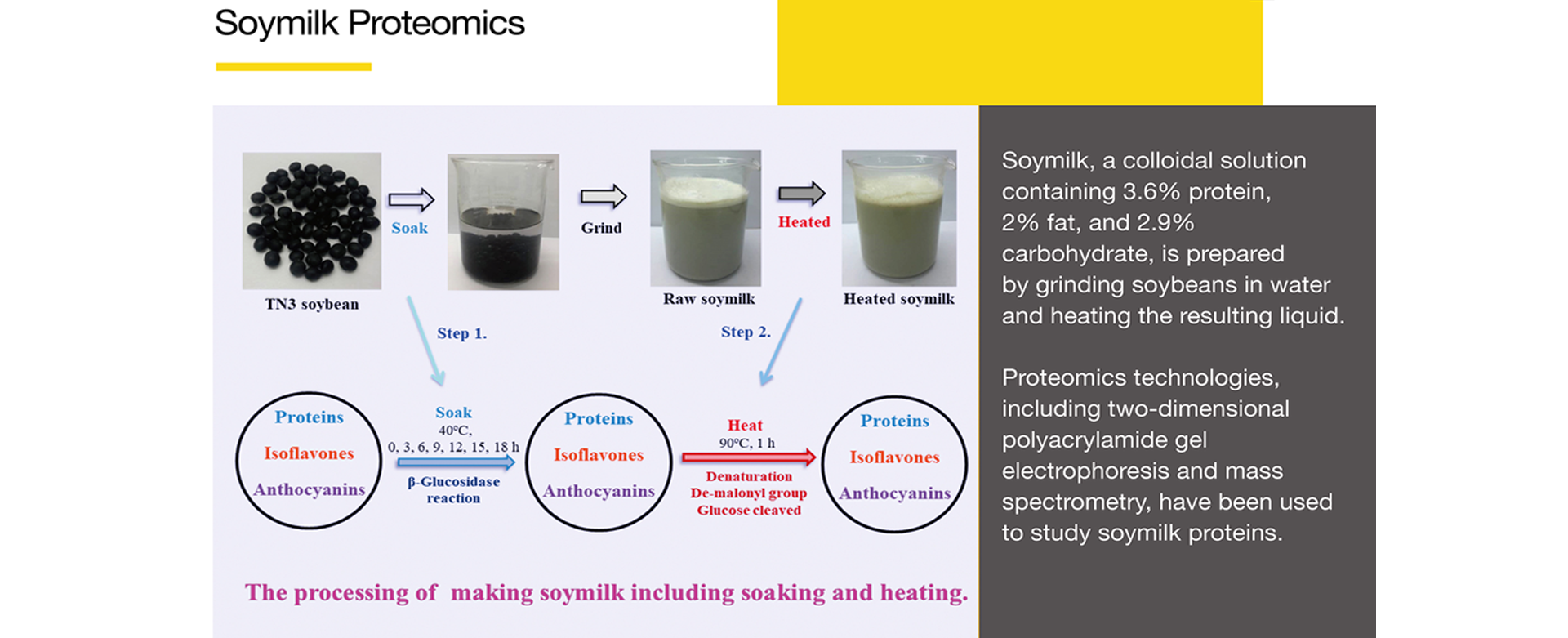
Soymilk, a colloidal solution containing 3.6% protein, 2% fat, and 2.9% carbohydrate, is prepared by grinding soybeans in water and heating the resulting liquid.
Proteomics technologies, including two-dimensional polyacrylamide gel electrophoresis and mass spectrometry, have been used to study soymilk proteins.
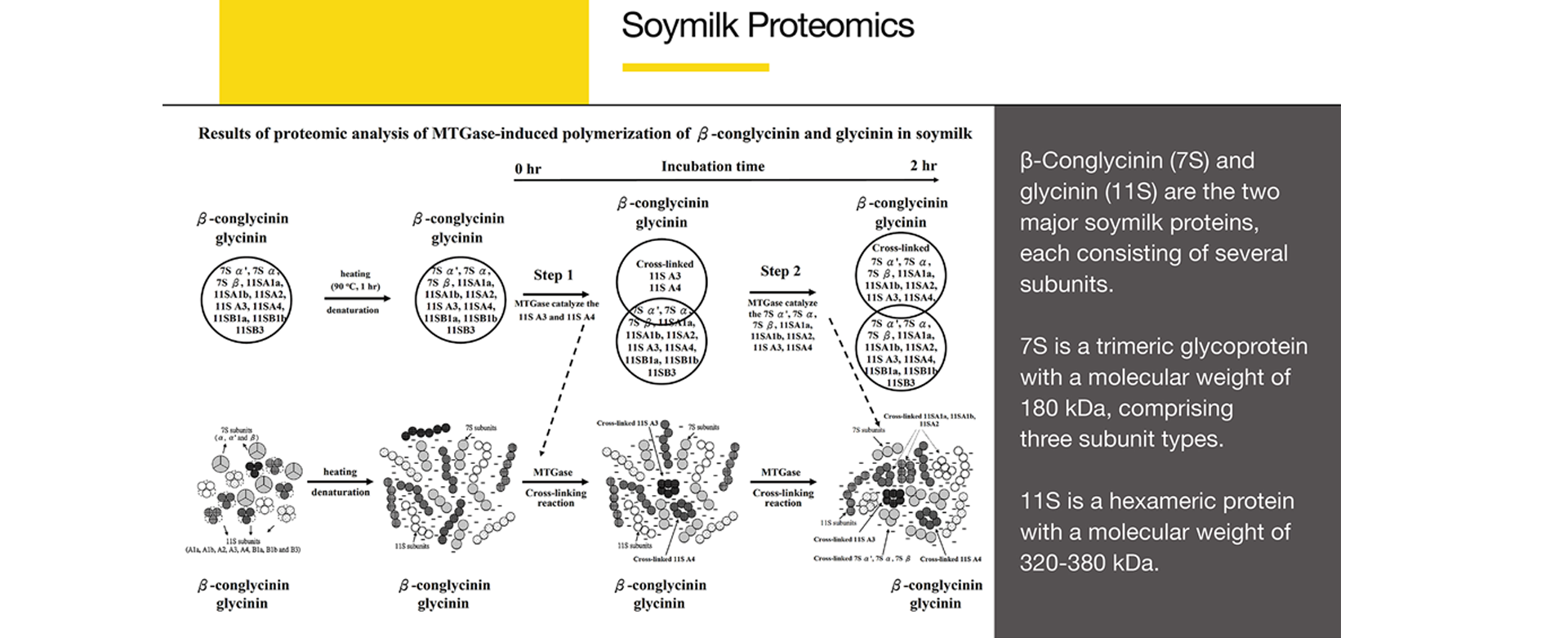
β-Conglycinin(7S) and glycinin(11S) are the two major soymilk proteins, each consisting of several subunits.
7S is a trimeric glycoprotein with a molecular weight of 180 kDa, comprising three subunit types.
11S is a hexameric protein with a molecular weight of 320-380 kDa.
Eggs are a rich source of proteins. These proteins provide several functions. The primary purpose of egg white is to protect the yolk and provide additional nutrition for the growth of the embryo.
Proteomic study of egg white can help us understand the secrets of the egg white proteins.
134 views